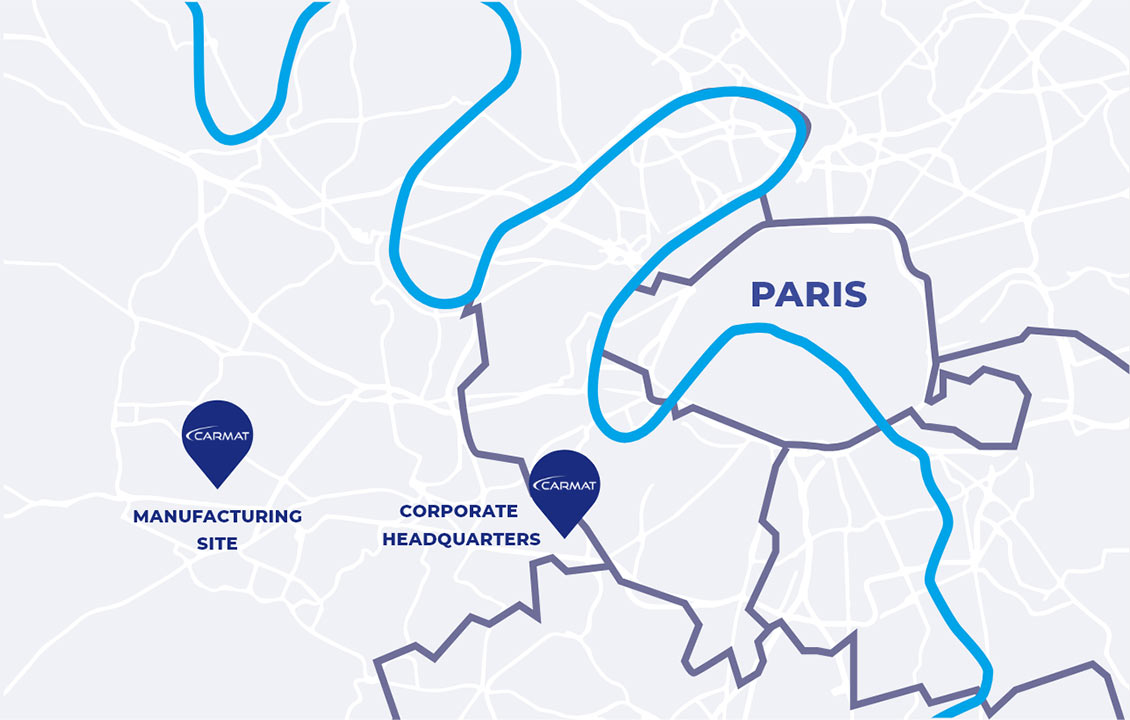


Carmat
Become the primary alternative to heart transplants with the implementation of our Total Artificial Heart.
Carmat will provide cardiologists with innovative technologies that save lives and improve the quality of life for patients with advanced heart failure.
2024
2023
2022
26/12/2022
First implant of Aeson® within the framework of the EFICAS clinical study in France. This prospective study will involve a total of 52 patients eligible for a heart transplant in France and will allow CARMAT to collect both additional data on the efficacy and safety of its artificial heart and medico-economic data to support its value proposition and the device’s reimbursement, notably in France. The study’s primary endpoint is survival for 180 days after implantation of the device without a disabling stroke, or a successful heart transplant within 180 days of implantation.
2021
15/07/2021
First human implant of Aeson® total artificial heart in the United States within the framework of the Early Feasibility Study (EFS). In accordance with the study protocol approved by the FDA, 10 transplant-eligible patients are expected to be enrolled in this trial. The primary study endpoint is patient survival at 180 days post-implant or a successful cardiac transplantation within 180 days post-implant. It is a staged study with a progress report of the first 3 patients after 60 days, before the enrollment of the next 7 patients.
2020
12/10/2020
The French National Authority for Health (HAS) confirmed its positive opinion regarding the financing by special exemption of the CARMAT total artificial heart within the framework of the EFICAS study, a prospective, multicenter, non-randomized study to be undertaken on 52 patients in France. Carmat has been granted €13 million in funding by the French Ministry of Health and Solidarity to conduct the EFICAS clinical study.
05/02/2020
Carmat received full approval from the FDA (Food & Drug Administration) to undertake a clinical feasibility study in the United States on 10 patients eligible for a transplant. The Centers for Medicare & Medicaid Services (CMS) approved coverage of the Carmat device and routine care items and services supplied to patients within the framework of this study.
2018
2017
31/12/2017
Applying for CE marking and commercial authorization. The CE mark is a prerequisite to market the prosthesis in Europe. It entails conformity with the EU regulations regarding design, manufacturing and clinical evaluation. The objective of the pivotal study is to demonstrate safety and performance of the Carmat TAH.
2016
2013
2011
2008
Carmat governance ensures an effective and transparent organization for its different supervisory and operational management teams to create long-term value for all stakeholders. It includes the Management Team and the Board of Directors.
The team focuses on implementing the company’s strategy, overseeing operations, designing action plans, measuring progress, reviewing success in meeting targets, managing change and spearheading the R&D strategy.

STÉPHANE PIAT
Chief Executive Officer
Stéphane Piat is an acknowledged specialist in the medical device business, particularly in the field of cardiology. He joined Carmat as C...

STÉPHANE PIAT
Chief Executive Officer
Stéphane Piat is an acknowledged specialist in the medical device business, particularly in the field of cardiology. He joined Carmat as Chief Executive Officer in September 2016.
Mr Piat started his career at Becton Dickinson European Headquarters as a Market Researcher in 1995. He was appointed European Platform Leader for Locoregional Anaesthesia five years later. In 2002, he joined Cordis, a Johnson & Johnson company, where he spent five years in several management positions ranging from Business Director France to European Marketing Director for Cardiology. In 2007, he moved to Abbott Vascular as General Manager for mid-size countries, EMEA, and two years later oversaw the integration of Evalve as the company’s General Manager EMEA, heading clinical and commercial development of a new interventional cardiology product, Mitraclip. In 2014, he led Global Market Development of the Abbott Vascular Structural Heart Division in San Francisco as Division Vice President.
Mr Piat holds a master’s degree in Management Science from IAE Dijon School of Management, and a post-graduate degree in Quantitative Marketing from ESA business school in Grenoble.

PASCALE D'ARBONNEAU
Deputy Chief Executive Officer & Chief Financial Officer
A graduate of the ESCP Europe business school, Pascale d’Arbonneau is a member of the Association nationale des directeurs financiers et ...

PASCALE D'ARBONNEAU
Deputy Chief Executive Officer & Chief Financial Officer
A graduate of the ESCP Europe business school, Pascale d’Arbonneau is a member of the Association nationale des directeurs financiers et de contrôle de gestion, (DFCG, the French association of financial and management control managers) and a lecturer at Paris Diderot University. Before joining CARMAT, Pascale d’Arbonneau was Executive Director of the Econocom International B.V. (EIBV) family office.
She began her career in 1989 as an auditor at Coopers & Lybrand (now PWC) before entering the pharmaceutical industry as Head of Finance & IT France at Johnson & Johnson – MSD (1995-1999). She spent most of her career (1999-2016) at GlaxoSmithKline (GSK). She joined as Director, Head of Controlling & Finance Partnering, France at GlaxoWellcome, just before its merger with SmithKline Beecham, managing a team of 20 people working on the combination of the two companies. She then held a number of senior positions within the Group (Vice President & Finance Controller, Pharma Europe from 2006 to 2010, Vice President & Area Finance Director, Western Europe from 2010 to 2014) before becoming Vice President Compliance and Control Integration for all business units worldwide.

MARC GRIMMÉ
Director of Research & Development
Since 1996, Marc Grimmé has been the technical lead on the programme to develop the Carmat bioprosthetic heart.

MARC GRIMMÉ
Director of Research & Development
Since 1996, Marc Grimmé has been the technical lead on the programme to develop the Carmat bioprosthetic heart.
He began his career in 1991 at MBDA France, where he worked on a range of issues linked to the development of mission-critical electronics, from upstream studies and the design phase to production commissioning.
Marc Grimmé is a graduate of the Institute Supérieur d’Electronique et du Numérique (ISEN).

PR CHRISTIAN LATREMOUILLE
Chief Medical Officer
Doctor of Medicine specialized in Heart Surgery, Christian Latrémouille is a Professor at Paris University, formed by the recent merger of...

PR CHRISTIAN LATREMOUILLE
Chief Medical Officer
Doctor of Medicine specialized in Heart Surgery, Christian Latrémouille is a Professor at Paris University, formed by the recent merger of Paris-Descartes (Paris V) and Paris-Diderot (Paris VII) Universities.
Before joining CARMAT, Christian Latrémouille was, since 2017, Head of the Cardiovascular Surgery Department at the Georges Pompidou European Hospital in Paris. He began his career in 1993 as Senior Specialist Registrar within Prof. Alain Carpentier’s prestigious Cardiac Surgery department at the Broussais Hospital in Paris. Upon validation of a PhD in xenotransplantation, in 1995 he took charge of the heart transplant program. Initially Clinical Lecturer in 1995 and then University Lecturer in 2000, in 2004 he was appointed Associate Professor at Paris-Descartes University, university chair of Clinical Anatomy and hospital chair of adult Cardiac Surgery. Since then, he has been entrusted with the preclinical development phase of the CARMAT bioprosthetic total artificial heart.

RAOUIA BOUYANZER
Human Resources Director
Raouia has nearly 16 years of human resources and payroll experience. Her broad experience includes benefits, employee relations, payroll, ...

RAOUIA BOUYANZER
Human Resources Director
Raouia has nearly 16 years of human resources and payroll experience. Her broad experience includes benefits, employee relations, payroll, talent acquisition and training. She started her career in 1998 in an accounting firm. In 2001, Raouia joined Morgan Stanley, where she held positions in social benefits management audit, payroll and human resources for more than 9 years. Raouia joined CARMAT at the stage of the ‘development’ of the company in February 2011 as an Administrative and Financial Manager, and set up in 2012 the human resources policy.
Raouia is a graduate of ESSEC business school in Human Resources Management and holds a Financial and Accounting degree (2001).

FRANCESCO ARECCHI
Director of Global Market Development
A marketing professional with strong experience in global leading companies within the healthcare industry, Francesco Arecchi joins Carmat ...

FRANCESCO ARECCHI
Director of Global Market Development
A marketing professional with strong experience in global leading companies within the healthcare industry, Francesco Arecchi joins Carmat in September 2017. Francesco Arecchi spent most of his career in Life Sciences companies such as Johnson & Johnson and Abbott, where he holds a number of positions from sales to marketing in Cardiology breakthrough technology products such as Cypher and MitraClip.
Prior to joining Carmat, he stood as Product Manager EMEA Structural Heart at Abbott. Francesco Arecchi is a biomedical engineer and graduated from Politecnico di Milano (Italy) with an MBA from Rotterdam School of Management (Netherlands).

THIERRY DUPOUX
Senior Director of Quality Assurance
Thierry Dupoux is a seasoned medical device professional with a strong and large expertise in Quality Assurance/Regulatory Affairs and R&D....

THIERRY DUPOUX
Senior Director of Quality Assurance
Thierry Dupoux is a seasoned medical device professional with a strong and large expertise in Quality Assurance/Regulatory Affairs and R&D.
Engineering Graduate from Ecole Centrale de Lyon (France), he has worked most of his career for Life Sciences companies such as General Electric where he became Supply Chain Quality & Compliance Manager for the plant of Buc (France) in his last position. In 2006, he joined Sorin Group, now named LivaNova, a world leader in Cardiac Surgery and Neuromodulation.
Over the past 12 years at LivaNova, he held several senior positions in Quality Assurance, Regulatory Affairs and R&D. Prior to joining CARMAT, he was Vice President of Quality Assurance at LivaNova where he led the integration of the Quality Systems following the merger between Sorin Group and Cyberonics.
The Carmat Board of Directors comprises 9 members (including 4 independent members) who bring a wide array of complementary professional experience to the table. The Board of Directors meets as often as is needed in the interests of the company and at least once every three months, in line with statutory guidelines. Members are appointed for a renewable period of 3 years. The Board of Directors determines the strategic, economic, social, financial and scientific focus of Carmat’s activities and ensures that relevant initiatives are implemented.

PIERRE BASTID
Chairman
Pierre Bastid has spent more than 25 years in senior executive positions at major international business groups such as Schlumberger, Schne...

PIERRE BASTID
Chairman
Pierre Bastid has spent more than 25 years in senior executive positions at major international business groups such as Schlumberger, Schneider Electric, Valeo and Thomson. Former President and CEO of Converteam Group, acquired from Alstom, he developed and sold to General Electric after the value increased by 30. His fund ZAKA has especially invested in several biotechnology companies including Carmat and Cellectis of which he is Board member.
He first joined the Carmat Board of Directors on 5 April 2018.

PROFESSEUR ALAIN CARPENTIER
Honorary President
Professor Emeritus at Descartes University in Paris and adjunct professor at Mount Sinai Medical School in New York, Alain Carpentier has m...

PROFESSEUR ALAIN CARPENTIER
Honorary President
Professor Emeritus at Descartes University in Paris and adjunct professor at Mount Sinai Medical School in New York, Alain Carpentier has made decisive contributions to the field of surgery for heart valves and heart failure. He is considered by many in the international community to be the founding father of modern valve surgery, after breaking with the traditional use of mechanical prostheses to develop biological and physiological solutions to problems related to heart valve replacement.
He was awarded the Grand Prize by the French Foundation for Medical Research and in 2007 received the prestigious Albert Lasker Medical Research Award for the invention of bioprostheses and the development of plastic/reconstructive surgery for heart valves.
Professor Carpentier also holds a range of responsibilities as Chairman of the Scientific Committee at Fondation Lefoulon-Delalande Institut de France, Member of the Board at Fondation Singer Polignac, and Director of the Alain Carpentier Foundation Scientific Research Association (ARSFAC).
Professor Carpentier first joined the Carmat Board of Directors on 7 May 2010.

JEAN-LUC LEMERCIER
Independent Director
Jean-Luc Lemercier draws on more than 20 years' experience and acknowledged leadership in medical devices. During his career, he has held a...

JEAN-LUC LEMERCIER
Independent Director
Jean-Luc Lemercier draws on more than 20 years’ experience and acknowledged leadership in medical devices. During his career, he has held a number of key positions in the field of cardiology, notably at Johnson & Johnson Cordis from 1996 to 2008, where he created and headed the Structural Heart Disease division. Since 2008, he has been Corporate Vice President EMEA, Canada & Latin America at Edwards Lifescience.
Mr Lemercier graduated in pharmacy from Claude Bernard Lyon 1 University.
He first joined the Carmat Board of Directors on 24 April 2017.

PAOLO RAMPULLA
Director Representing Santé Holdings
Paolo Rampulla is a well-known tax specialist, notably in relation to Mergers & Acquisitions, « private equity », international taxation ...

PAOLO RAMPULLA
Director Representing Santé Holdings
Paolo Rampulla is a well-known tax specialist, notably in relation to Mergers & Acquisitions, « private equity », international taxation et taxation of financial instruments, particularly in the healthcare & life sciences, automotive and oil & gas sectors.
An Italian citizen, Paolo Rampulla holds a Master in Tax Law from Ipsoa School (Milan, Italy) and a degree in Economics from the University Luigi Bocconi (Milan, Italy).

DR. MICHAEL MACK
Independent Director
Michael Mack is an internationally renowned cardiac surgeon with extensive experience in the introduction of medical devices and innovative...

DR. MICHAEL MACK
Independent Director
Michael Mack is an internationally renowned cardiac surgeon with extensive experience in the introduction of medical devices and innovative procedures for cardiovascular disease. He has authored more than 500 scientific publications and has received the Presidential Citation from the American College of Cardiology (ACC) and the Transcatheter Cardiovascular Therapeutics (TCT) Lifetime Achievement Award.
Dr Mack is a graduate of Boston College, St Louis University and the University of Texas Southwestern Medical School. He is also the Director of the Cardiovascular department for pharmaceutical firm Baylor Scott & White Health, a Director on the American Board of Thoracic Surgery and a member of the FDA Medical Device Epidemiology Network Initiative (MDEpiNet) Advisory Committee.
He first joined the Carmat Board of Directors on 24 April 2017.

STÉPHANE PIAT
Chief Executive Officer
Stéphane Piat is an acknowledged specialist in the medical device business, particularly in the field of cardiology. He joined Carmat as C...

STÉPHANE PIAT
Chief Executive Officer
Stéphane Piat is an acknowledged specialist in the medical device business, particularly in the field of cardiology. He joined Carmat as Chief Executive Officer in September 2016.
Mr Piat started his career at Becton Dickinson European Headquarters as a Market Researcher in 1995. He was appointed European Platform Leader for Locoregional Anaesthesia five years later. In 2002, he joined Cordis, a Johnson & Johnson company, where he spent five years in several management positions ranging from Business Director France to European Marketing Director for Cardiology. In 2007, he moved to Abbott Vascular as General Manager for mid-size countries, EMEA, and two years later oversaw the integration of Evalve as the company’s General Manager EMEA, heading clinical and commercial development of a new interventional cardiology product, Mitraclip. In 2014, he led Global Market Development of the Abbott Vascular Structural Heart Division in San Francisco as Division Vice President.
Mr Piat holds a master’s degree in Management Science from IAE Dijon School of Management, and a post-graduate degree in Quantitative Marketing from ESA business school in Grenoble.

David Coti
Independent Director
David Coti, 38, holds a double degree in international business (ESSEC International and Plekhanov University in Moscow).

David Coti
Independent Director
David Coti, 38, holds a double degree in international business (ESSEC International and Plekhanov University in Moscow). He started his career in the 2000s by creating a distribution company in the CIS (Commonwealth of Independent States). As an investor particularly interested in emerging markets, life sciences, biotechnologies and “clean technologies”, he has been managing since 2015 various family offices, including those of the Gaspard family (Bratya SPRL and Corely SPRL), owner of the Lyreco group. He is also, since 2016, Vice President Marketing of TBR, a company specialised in digital marketing. Finally, he is a member of the governance committee of Investir &+, an investment structure that supports the growth of entrepreneurs developing projects with a strong social or environmental impact.

Florent Battistella
Independant Director
Florent Battistella, 60, is a graduate engineer from INSA Toulouse and holds a PhD in solid state physics from Paul Sabatier University in ...

Florent Battistella
Independant Director
Florent Battistella, 60, is a graduate engineer from INSA Toulouse and holds a PhD in solid state physics from Paul Sabatier University in Toulouse. From 1983 to 1988, he held research positions at the CNRS in France and at the University of Cambridge in the United Kingdom. From 1988 to 2004, he worked in production at IBM (semiconductor manufacturing), then in the automotive sector at Valéo, and finally at Solectron (electronics manufacturing) where he was Vice President Operations, in charge of 9 European sites. From 2004 to 2011, he was Vice-CEO, then Chief Operating Officer and finally CEO of emerging countries at Converteam, a company under LBO acquired by General Electric in 2011. He then founded Nisima, a holding company, which holds stakes in various firms, notably in the naval and aeronautical sectors.

Laurent Kirsch
Chairman of the Audit Committee and Independant Director representing Thérabel Invest
Laurent Kirsch is an acknowledged expert in the healthcare sector, internationally. He also brings to the Board his significant experience ...

Laurent Kirsch
Chairman of the Audit Committee and Independant Director representing Thérabel Invest
Laurent Kirsch is an acknowledged expert in the healthcare sector, internationally. He also brings to the Board his significant experience on financial matters and as director of a number of other organizations.
Laurent Kirsch is currently Chief Executive Officer of THERABEL PHARMA group. Prior to this, he had held different senior positions within the ABBOTT group, including President of the French affiliate, Head (administtrateur-délégué) of the Belgium & Luxembourg business, director of the « Primary Care » business-unit in Italy and commercial director, Europe. Before joining ABBOTT, he also had roles of increasing responsibility at SCHERING-PLOUGH (now MERCK Inc.) and DELOITTE.
Laurent Kirsch holds a MBA (economics and finance) from Haute Ecole « Groupe ICHEC – Saint-Louis – ISFSC », Brussels.
The committee reviews the preparation and control of financial and accounting information in order to secure the accuracy of information communicated to investors. The committee reviews the conclusions of the works performed by external auditors on the financial information prepared by the company, including the audit of the financial statements and the assessment of internal control.

Laurent Kirsch
Chairman of the Audit Committee and Independant Director representing Thérabel Invest
Laurent Kirsch is an acknowledged expert in the healthcare sector, internationally. He also brings to the Board his significant experience ...

Laurent Kirsch
Chairman of the Audit Committee and Independant Director representing Thérabel Invest
Laurent Kirsch is an acknowledged expert in the healthcare sector, internationally. He also brings to the Board his significant experience on financial matters and as director of a number of other organizations.
Laurent Kirsch is currently Chief Executive Officer of THERABEL PHARMA group. Prior to this, he had held different senior positions within the ABBOTT group, including President of the French affiliate, Head (administtrateur-délégué) of the Belgium & Luxembourg business, director of the « Primary Care » business-unit in Italy and commercial director, Europe. Before joining ABBOTT, he also had roles of increasing responsibility at SCHERING-PLOUGH (now MERCK Inc.) and DELOITTE.
Laurent Kirsch holds a MBA (economics and finance) from Haute Ecole « Groupe ICHEC – Saint-Louis – ISFSC », Brussels.

David Coti
Independent Director
David Coti, 38, holds a double degree in international business (ESSEC International and Plekhanov University in Moscow).

David Coti
Independent Director
David Coti, 38, holds a double degree in international business (ESSEC International and Plekhanov University in Moscow). He started his career in the 2000s by creating a distribution company in the CIS (Commonwealth of Independent States). As an investor particularly interested in emerging markets, life sciences, biotechnologies and “clean technologies”, he has been managing since 2015 various family offices, including those of the Gaspard family (Bratya SPRL and Corely SPRL), owner of the Lyreco group. He is also, since 2016, Vice President Marketing of TBR, a company specialised in digital marketing. Finally, he is a member of the governance committee of Investir &+, an investment structure that supports the growth of entrepreneurs developing projects with a strong social or environmental impact.
The committee makes recommendations to the Board of Directors regarding potential appointments to the Management Team, the Board of Directors and key Carmat positions related to company governance. It also sets and reviews compensation policies for executive corporate officers and high-potential employees in line with the company’s targets and individual and collective performance.

JEAN-LUC LEMERCIER
Chairman
Jean-Luc Lemercier draws on more than 20 years' experience and acknowledged leadership in medical devices. During his career, he has held a...

JEAN-LUC LEMERCIER
Chairman
Jean-Luc Lemercier draws on more than 20 years’ experience and acknowledged leadership in medical devices. During his career, he has held a number of key positions in the field of cardiology, notably at Johnson & Johnson Cordis from 1996 to 2008, where he created and headed the Structural Heart Disease division. Since 2008, he has been Corporate Vice President EMEA, Canada & Latin America at Edwards Lifescience.
Mr Lemercier graduated in pharmacy from Claude Bernard Lyon 1 University.
He first joined the Carmat Board of Directors on 24 April 2017.

PAOLO RAMPULLA
Director Representing Santé Holdings
Paolo Rampulla is a well-known tax specialist, notably in relation to Mergers & Acquisitions, « private equity », international taxation ...

PAOLO RAMPULLA
Director Representing Santé Holdings
Paolo Rampulla is a well-known tax specialist, notably in relation to Mergers & Acquisitions, « private equity », international taxation et taxation of financial instruments, particularly in the healthcare & life sciences, automotive and oil & gas sectors.
An Italian citizen, Paolo Rampulla holds a Master in Tax Law from Ipsoa School (Milan, Italy) and a degree in Economics from the University Luigi Bocconi (Milan, Italy).
Download the Code of ethics and business conduct of Carmat